I found out at the beginning of this month that my internship was going to end a little sooner than I had originally planned on. I was supposed to stay here at Dorena until the end of March, but a new job offer shortened that timeline by a month. Now I sit here during my last week of work thinking of all the things I did and friends I made this winter.
I was lucky to not only be partnered with such an amazing mentor (s/o to Lee), but it turns out my co-intern ended up being my long lost twin. It became a joke at Dorena that wherever one of us was, the other wasn’t far behind. Whether it was during work or our personal time, I always knew that if I asked Brenn if she wanted a lil treat she would say yes and off we’d go. I could also always count on her for weekend adventures, whether it was a day at the beach, or going hiking no matter the weather, I knew Brenn would be down for anything.

Another major part of this internship for me was Teo, the grumpy nursery cat. I’ve talked about him in blogs before, but I love this cat. It has been so fun getting to know him (i.e. bothering him), and pretending like he loves me too. Recently, I’ve been trying to “train” him into letting me pick him up without scratching my eyes out. It’s going pretty well, but I don’t think anyone is willing to take this task up after I leave so Teo will forget what it’s like to be picked up.
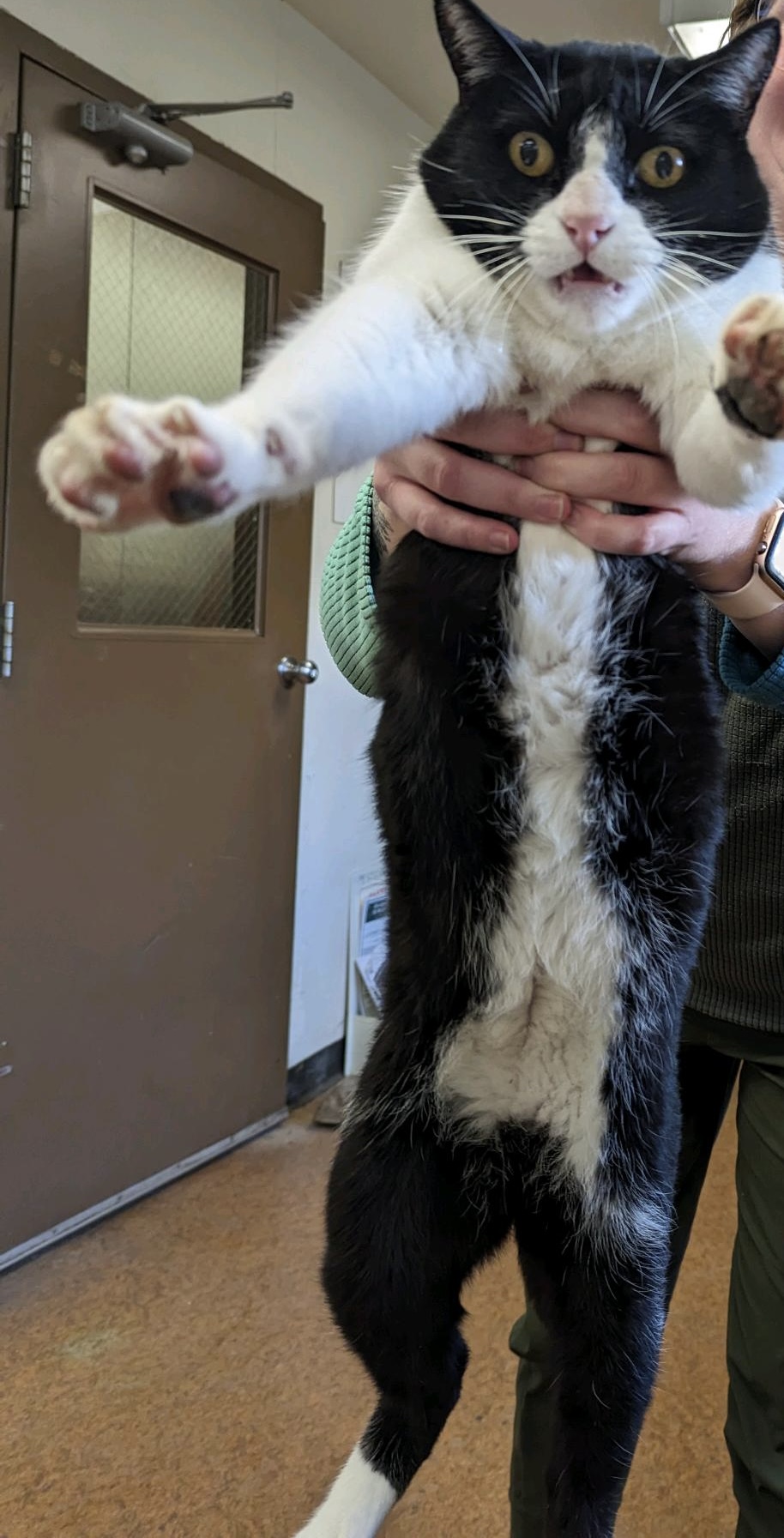
This last month has gone so fast, which is both good and bad. Good because I’m ready for the next adventure, but bad because I will miss the friends and co-workers I’ve made since being here. I’ll be working for USGS in Moab, Utah, so I’m very excited to get back to the desert and bask in the sun like the little lizard I am. One thing I have really loved about Oregon as a whole is the amount of rainbows I’ve seen since I’ve been here. They’re nice reminders that good things come out of rain, and the sun is just that much better after rain.














